 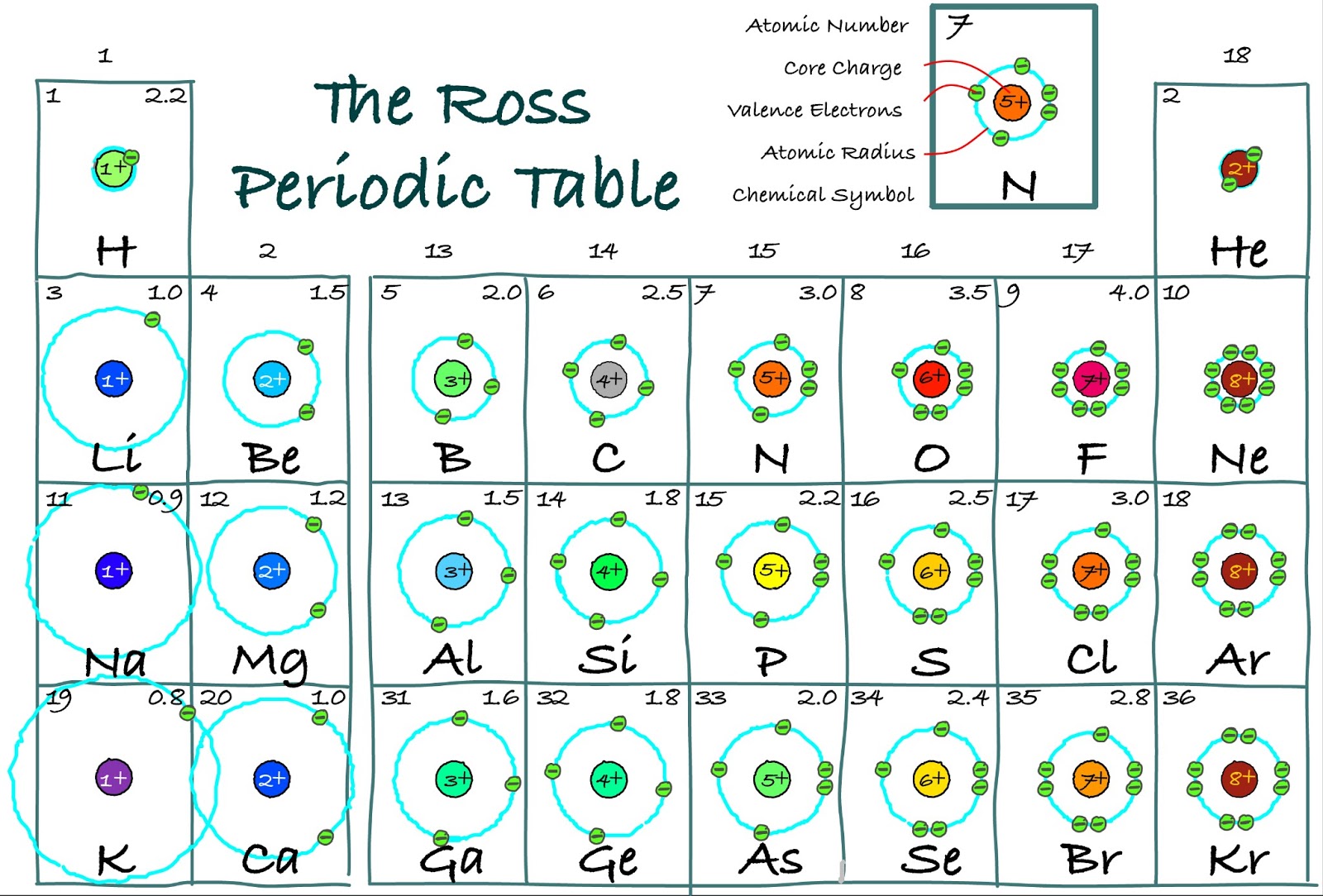
The atom with the larger coulombic value develops a negative charge due to having an excess of electrons. The electrons of the atom with the smaller coulombic value. When the two atoms come close and the coulombic attractions have a big difference (due to them being further away in the periodic table), the atom with the larger coulombic value steals When two atoms come close with different coulombic attractions, the atom with the larger coulombic attraction has the tendency to attract the electrons of the other atom which has a smaller coulombic attraction between its nucleus and electrons. Due to this, the size of the atom decreases along the period Coulombic Attraction in Bonding This is because the number of protons in the nucleus increases and thus the strength of the nucleus increases. Some atoms have a strong coulombic attraction compared to others due to the number of protons in the nucleus.Ītoms increase in coulombic attraction along the period. The strengths of the nuclei differ from atom to atom. Magnesium has a higher coulombic attraction than sodium.) Coulombic Attraction along the Periodĭue to the coulombic attraction, the electrons are attracted towards the positively charged nucleus which contains protons. (Sodium has a +1 charge and Magnesium has a +2 charge. This is why Magnesium ion is more reactive than Sodium Ion. Magnesium ion has a higher coulombic attraction compared to the coulombic attraction of Sodium. 
In magnesium, a larger charge is spread over the surface area compared to in sodium, where a smaller charge is spread over the surface area. However, both of them have the same number of electrons. The sodium ion has a charge of +1 and the magnesium ion has a charge of +2. Ions with a larger charge make stronger bonds compare to ions with a smaller charge. This is because the ion with a larger charge has more charge spread over a certain surface area and the ion with a lower charge has lower charge spread over a certain surface area. The ions with a larger charge will attract more opposite charged ions towards itself compared to ions with a smaller charge. The size of the charges on the ions makes a difference in the coulombic attraction. In ions, there is a coulombic attraction between the positively charged ions and negatively charged ion. Carbon, on the other hand, has electrons in the 1s2, 2s2 and 2p2 subshells.) (Lithium has electrons in the 1s2 and 2s1 subshells. The coulombic attraction is so high that Carbon is able to attract the electrons of other atoms giving it a -4 charge Say that Carbon has higher coulombic attraction compared to Lithium. Carbon attracts more electrons towards its nucleus compared to Lithium. Carbon has 6 protons whereas Lithium only has 3 protons. The increase in positive charge improves the strength of the nucleus and is able to pull the electrons which are even further away.Ĭarbon has more protons than Lithium. 
When there is a high number of protons, the positive charge increases. The size of the charge also affects the coulombic attraction. Sodium has 1s2 2s2 2p6 3s1 subshells occupied.) (Lithium only has 1s2 2s1 subshells occupied. This is also a reason why Li is more reactive than Na. That is why Li has a larger coulombic attraction compared to Na. The charge gets distributed over a larger surface area in Na compared to Li. Na 1 has a bigger atom and more electrons, but the charge is the same as Li 1+. Taking Li 1+ and Na 1+, both have the same charge, but the number of electrons and occupied shells is different. In a charged atom, the bigger the atom, the less is the coulombic attraction. 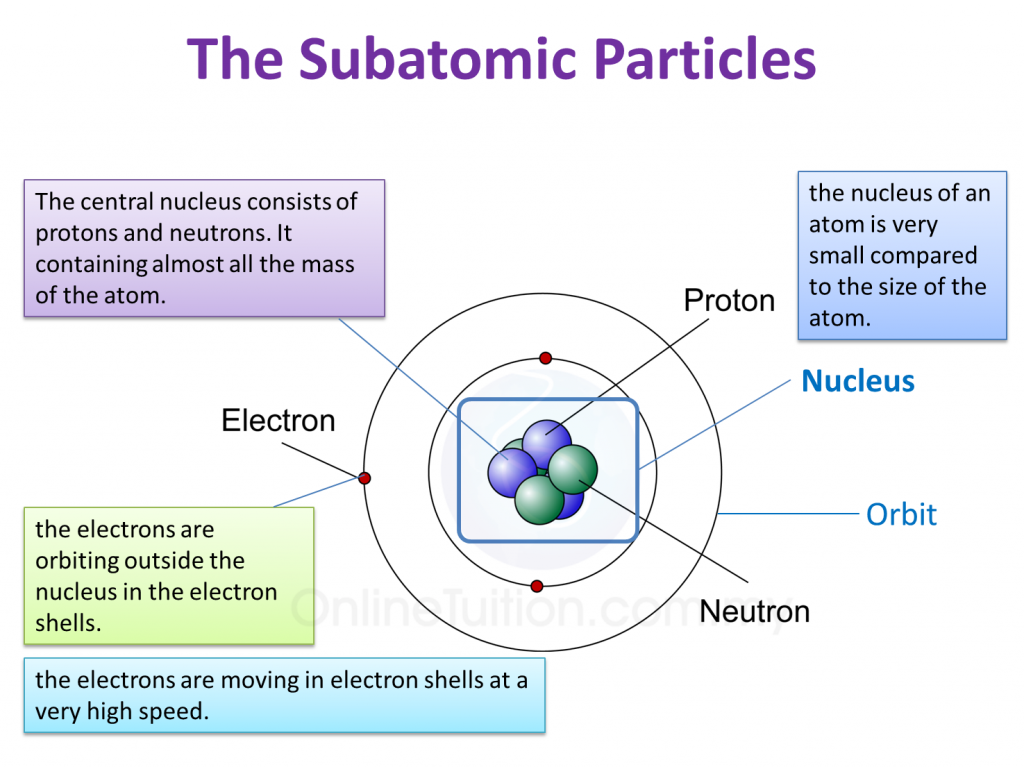
The nucleus is not able to pull the electrons, that are in orbitals further away from the nucleus, towards itself and the coulombic attraction decreases. The bigger the size of the atom, the electrons, especially the valence electrons are further away from the nucleus. The strength of the coulombic attraction depends on two things: This coulombic attraction causes electrons to orbit around the nucleus.) (The electrons are attracted to the nucleus because the nucleus has positively charged protons in it. positively charged ions and negatively charged ions being attracted to each.protons (which are positively charged) and electrons ( which are negatively charged) attracted to each.Similarly, oppositely charged particles are also attracted to each other. Like, the South Pole of a magnet and the North Pole of another magnet attract. Factors which affect the coulombic attraction Key definition of what Coulombic attraction isĬoulombic Attraction is the attraction between oppositely charged particles.Key points which are covered in this article: Coulombic Attraction in Covalent Bonding.Factors which affect the coulombic attraction. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
